With a steady increase of IBD prevalence worldwide and discovery of new disease pathways, we see the growing interest of pharmaceutical companies in IBD drug development. A survey of Trialtrove Informa database (Pharma Intelligence) in April 2019 revealed 47 companies conducting studies with more than 70 investigational or approved drugs in the IBD therapeutic area, composed of 43 compounds for UC, 29 for CD, and 6 for both indications [19]. During the 10-year period from 2010 to 2020, the number of clinical trials in the IBD therapeutic area almost doubled not only because of a continuous development of biologics, but also due to discovery of new pathways and consequently new classes of medications.
As we mentioned, IBD (both UC and CD) is a chronic inflammatory condition that will not be completely eliminated and thus will require a lifelong therapy. Commonly used, so called conventional therapy, including 5-aminosalicylates, corticosteroids, thiopurines, anti-TNFs and others, is generally effective in controlling symptoms and to some extent, pathological changes, it will not work if disease progresses. Such adverse side effects as immunosuppression and related to that systemic fungal infections, and potential for cancer development may have a negative impact on the disease outcome.
Currently, the drugs under investigation belong to 4 therapeutic classes with several mechanisms of action: monoclonal antibodies (α2β1 integrin antagonists, Interleukin-6/12/21/23 antagonists, CD40 and CD134 antagonists), protein kinase inhibitors (JAK 1/3 inhibitors), immunosuppressants (S1P receptor modulators), and antisense therapy (ICAM1 antagonists, TLR9 agonists).
In such competitive landscape with the large and growing number of compounds utilizing different mechanisms of action it is critical to understand how a potential drug may fit the major unmet needs in the IBD management.
Based on the literature analysis and the feedback from clinical trial investigators (and indirectly from the patients), we can conclude that the primary unmet need in management of moderate to severe IBD is lack of
oral drug formulations: patients would prefer an oral formulation over the subcutaneous or intravenous administration.
The first oral drug for UC was Pfizer’s Xeljanz (tofacitinib), a small molecule directed against the JAK/STAT pathway, blocking the inflammatory cascade, which received the FDA approval for this indication in 2018. The efficacy of Xeljanz for the treatment of moderately to severely active UC was demonstrated in three placebo-controlled clinical trials. Initially it was considered that the incidence of tofacitinib-related adverse events (AEs) in UC setting does not differ from that of patients with rheumatoid arthritis (the primary indication for this drug) and there were no particular safety concerns. However, after a 1 year of use in UC patients, the FDA issued a black box warning for the 10-mg, twice-daily dosage of tofacitinib in patients with
UC due to a potential for increased blood clot and death risks that were seen in a rheumatoid arthritis trial [20].
Recently, Bristol Myers Squibb announced detailed results from True North, a pivotal, placebo-controlled Phase 3 trial evaluating oral Zeposia (ozanimod), an oral, sphingosine 1-phosphate (S1P) receptor modulator that binds with high affinity to S1P receptors 1 and 5, as an induction and maintenance therapy in adult patients with moderate to severe ulcerative colitis (UC) who did not adequately respond to prior treatment [21]. The study demonstrating statistically significant and clinically meaningful results for clinical remission compared to placebo at induction and in maintenance. The overall safety observed was consistent with the known safety profile for Zeposia and patients with moderate to severe UC,
however, it is obvious that more safety data, and particularly the long-term ones, are necessary prior to recommending Zeposia for an extensive use. The first drug in this class, Gilenya (fingolimod), has been used in treatment of multiple sclerosis, and recently raised some concerns due to first-dose bradycardia and atrioventricular block. However, it will not be correct to directly extrapolate fingolimod safety data to ozanimod, as the adverse effects’ profile depends on the particular receptors targeted by the drug, with S1P2 and S1P3 receptors being associated with cardiovascular, pulmonary, and theoretical cancer-related risks, and targeting S1P1 receptors proved to be beneficial for inflammatory conditions. Fingolimod is a non-selective S1P modulator targeting S1P1, S1P3, S1P4, and S1P5 receptors, leading to a wide range of adverse events. Obviously the current
focus of drug development is on selective S1P modulators that targets predominantly or exclusively S1P1 receptors.
Until recently, in addition to ozanimod, two S1P modulators with differing selectivity for S1P receptors were in clinical development for IBD: etrasimod and amiselimod. Recently published results of a phase 2 placebo-controlled study of amiselimod in moderately to severe CD did not show a significant effect on clinical disease activity, however there were no clinically significant reports of bradycardia, ventricular tachycardia, or atrioventricular block, as well as no deaths, opportunistic infections, clinically relevant negative laboratory or abnormal ECG findings [22].
Phase 2 study data on etrasimod, a selective S1P1, S1P4, and S1P5 receptor modulator concluded that
the dose of 2 mg was more effective than placebo for improving the modified Mayo Clinical Score at week 12 in patients with moderately to severely active UC. However, the study has limitations: this was an induction study with only 12 weeks in duration and 156 patients randomized, and the safety and efficacy during longer-term maintenance therapy were not investigated [23]. The safety and efficacy of it will be further evaluated in phase 3 clinical studies.
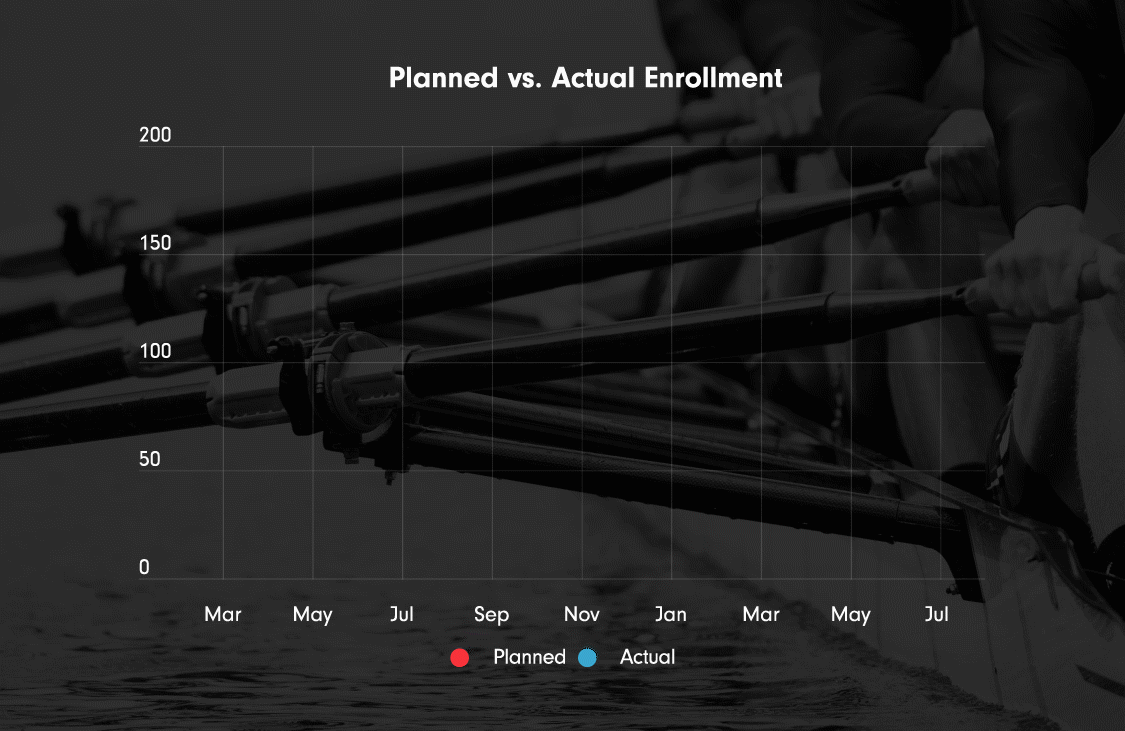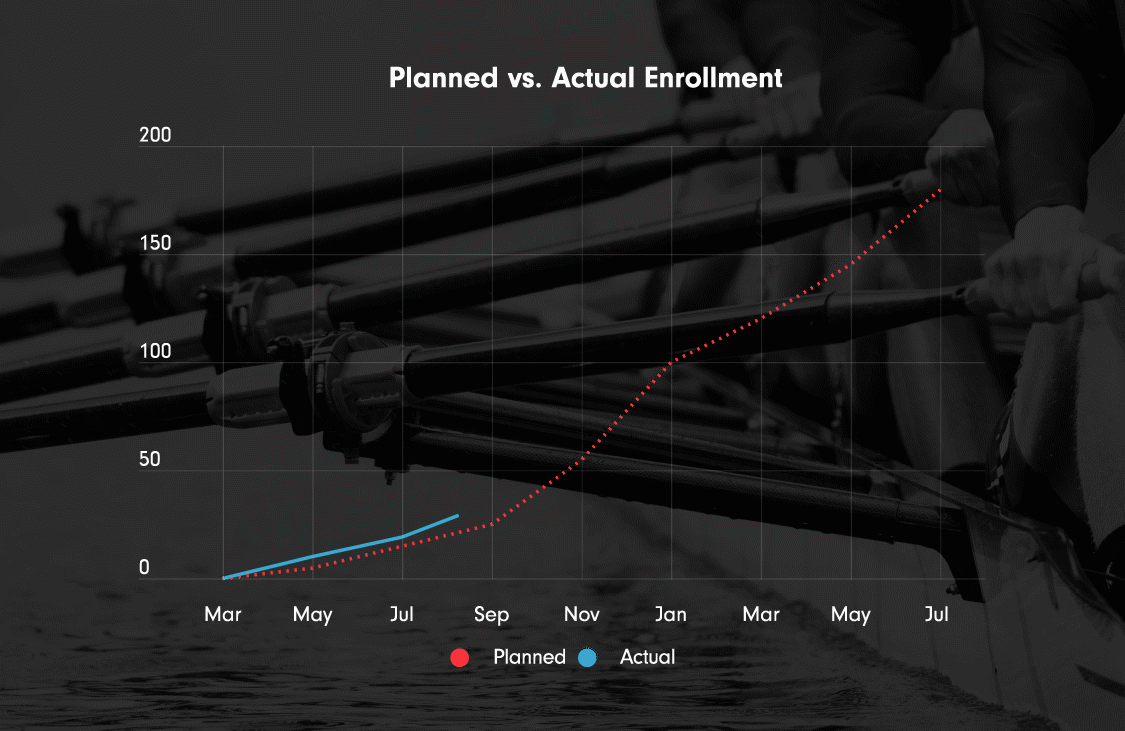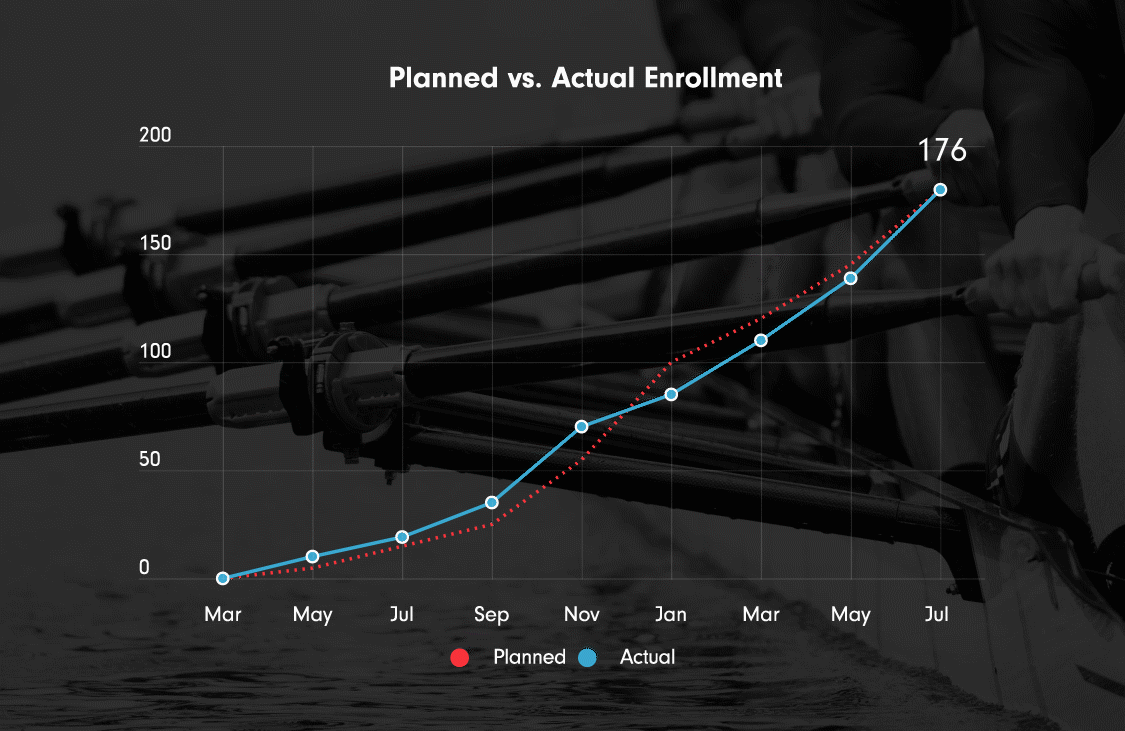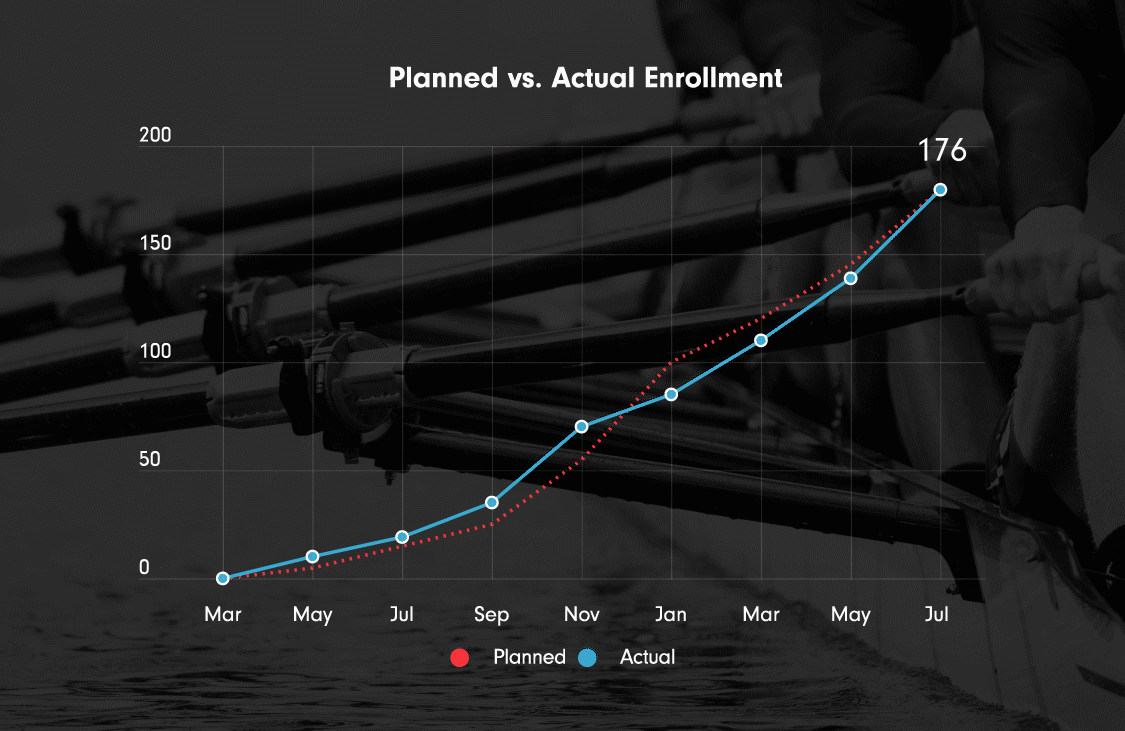
We may speculate that the growing number of new drugs is a good sign, which is definitely true, but there is the other side of the coin: the number of clinical trials in the IBD area does not go in parallel with the number of patients enrolled in the studies, and moreover, enrollment rate moves in the opposite direction showing a significant decline. For example, during a 20-year period from 1998
to 2018, the average recruitment rate in moderate-to-severe UC decreased from 0.32 to 0.13 patients per site per month, while the average recruitment rate in moderate-to-severe CD decreased from 0.65 to 0 to 0.10 patients per site per month [19]. Paradoxically, enrollment was influenced by success in drug development, particularly by the FDA approvals of vedolizumab in 2014, ustekinumab in 2016, and tofacitinib in 2018. With new therapies available, the interest to placebo-controlled trials (a frequent clinical trial design in the IBD setting) has decreased.

Ulcerative colitis, Phase III (ongoing)
medications), preferred study sites, with whom PSI has a long story of cooperation, and appropriately trained study teams. Appropriate selection of participating countries is now becoming one of the key points the incidence of IBD is increasing in Asia, Africa, and Latin America [1], making a logical movement of shifting clinical trials to these locations. Here PSI is in a pole position having not only well established operations in these areas, but also
pre-selected sites and trained personnel.
However, enrolling the required number of patient within an agreed time will be only part of the study (and sometimes not the biggest part). Over the past decade, screen failure rates in IBD trials have grown. Although actual numbers are not available, but according to the available literature sources, the screen failure rates in UC are now approaching 50% in UC and 70% in CD with the major reasons responsible for that being the failure to meet minimal endoscopic or biomarker criteria for active disease and growing list of exclusionary concomitant medications [19]. In PSI studies we’ve seen a screen out rate of 26-48% in UC and up to 64% in CD. There is no easy and universal solution of this problem, however general recommendations may include optimization of study outcomes, including endoscopic weighted inclusion and outcome criteria, and what we consider critically important – to make long-term extensions available to patients.
Both recruitment and retention of patients in a clinical trial depends on the study protocol criteria and complexity, which has to be scientifically sound on the one hand – to be able to achieve the goals of the study, and on the other hand – to be attractive to the patients. PSI has a solution for this task, providing a thorough review of the protocol by both internal therapeutic area experts and external independent consultants, who are not only key opinion leaders in the particular therapeutic area, but also principal investigators in clinical trials.
Crohn’s disease, Phase II
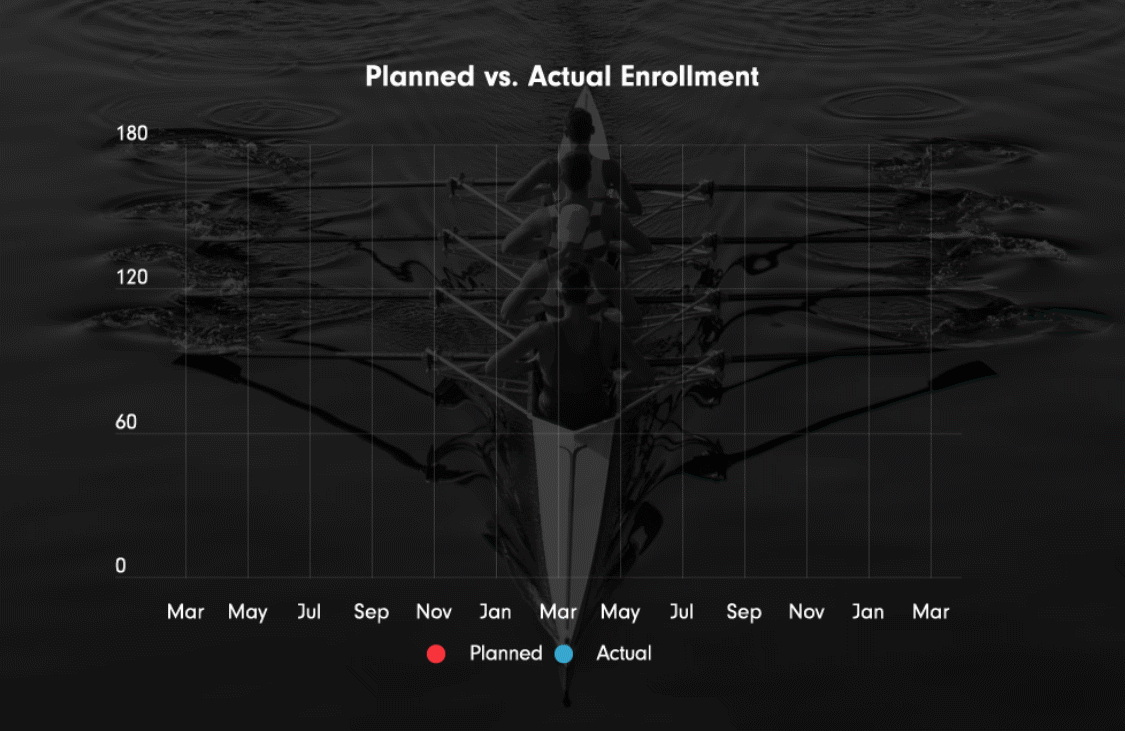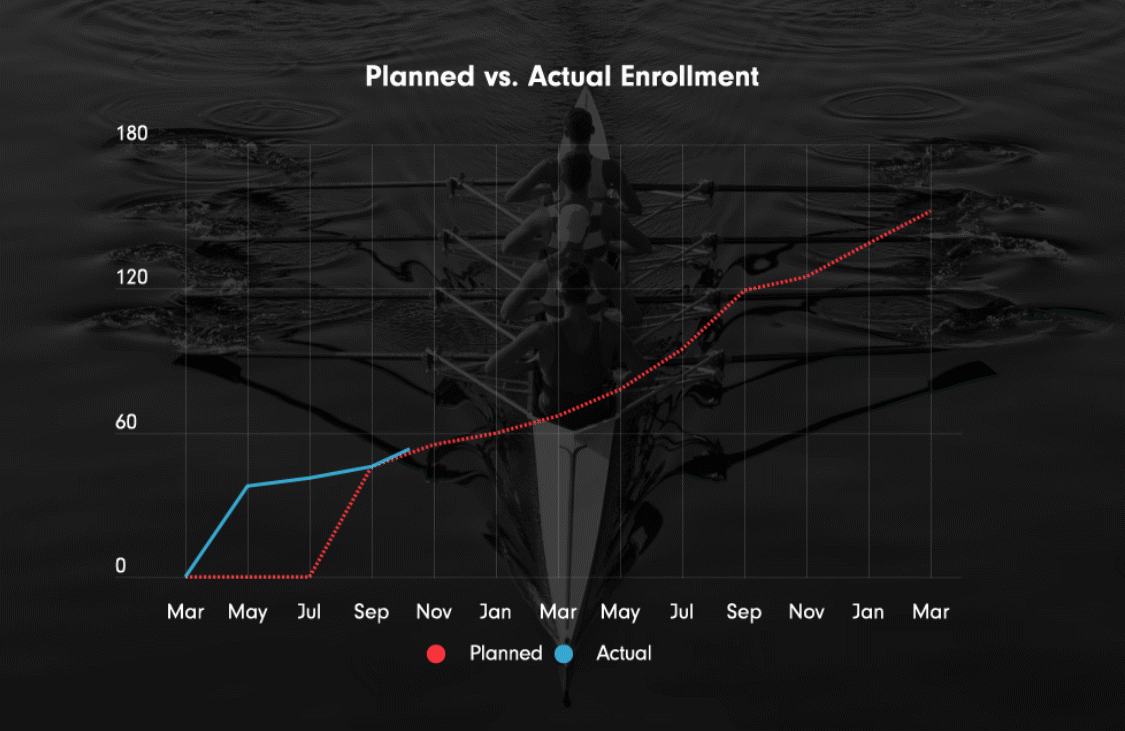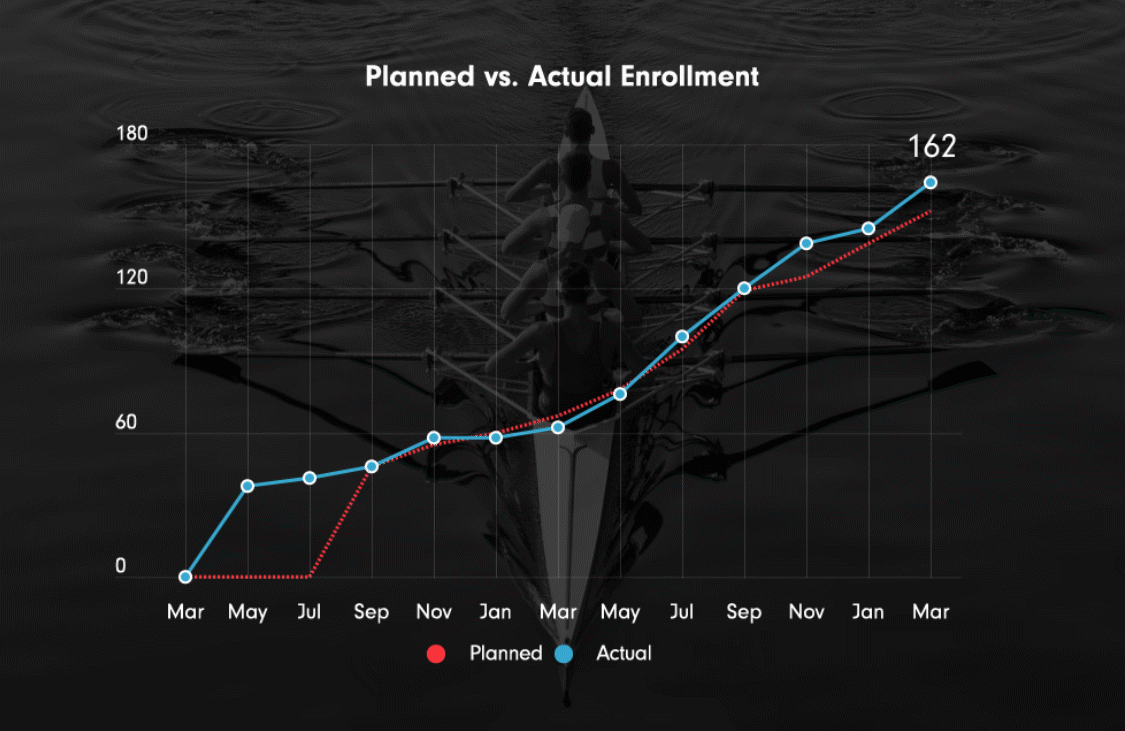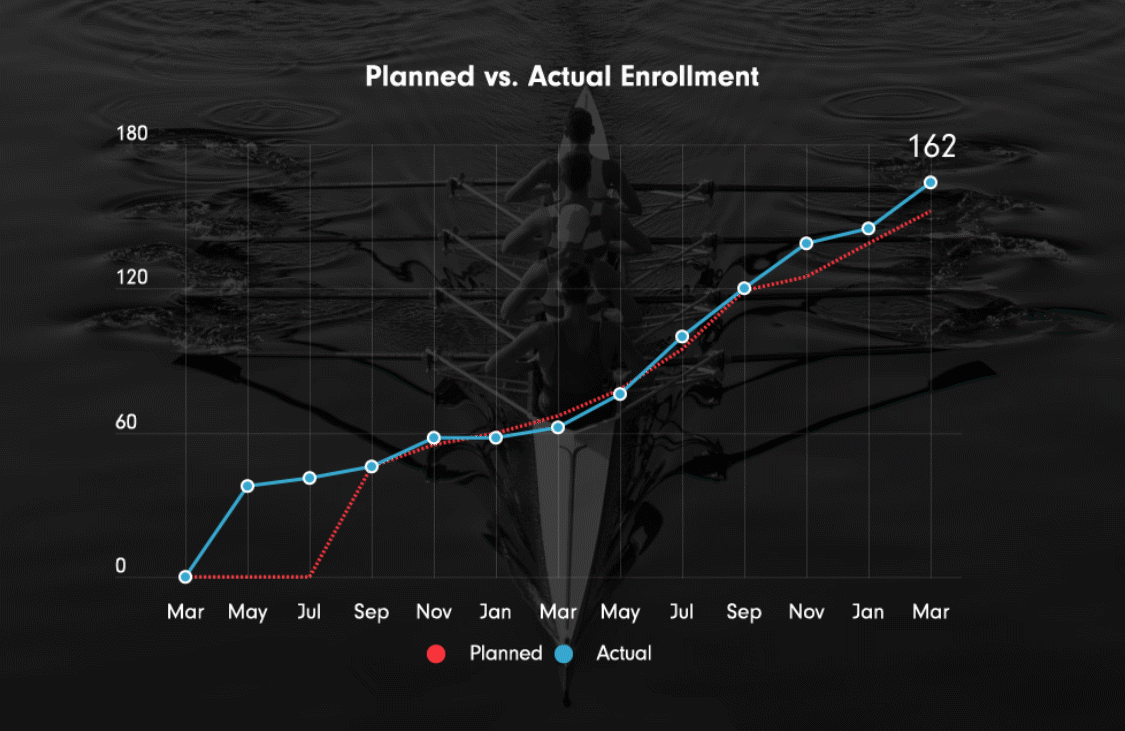
Ulcerative colitis, Phase III OLE extension (ongoing)